楊進木教授研究團隊發表研究成果於Front. Immunol.
連結網址:https://www.frontiersin.org/articles/10.3389/fimmu.2022.1080897/full
Abstract
Background: Drug repurposing is a fast and effective way to develop drugs for
an emerging disease such as COVID-19. The main challenges of effective drug
repurposing are the discoveries of the right therapeutic targets and the right
drugs for combating the disease.
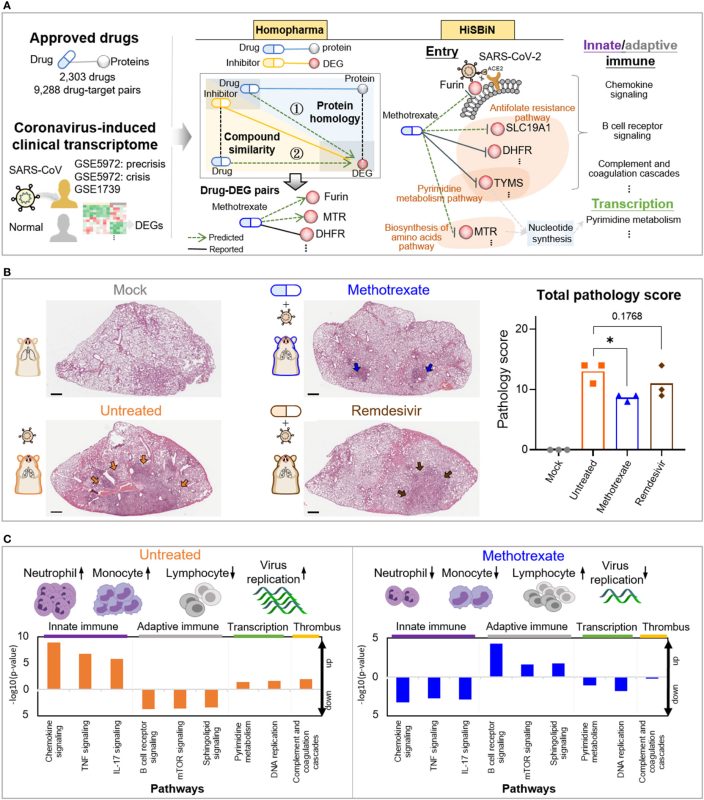
Methods: Here, we present a systematic repurposing approach, combining
Homopharma and hierarchal systems biology networks (HiSBiN), to predict 327
therapeutic targets and 21,233 drug-target interactions of 1,592 FDA drugs for
COVID-19. Among these multi-target drugs, eight candidates (along with
pimozide and valsartan) were tested and methotrexate was identified to
affect 14 therapeutic targets suppressing SARS-CoV-2 entry, viral replication, and COVID-19 pathologies. Through the use of in vitro (EC50 = 0.4 mM) and in
vivo models, we show that methotrexate is able to inhibit COVID-19 via
multiple mechanisms.
Results: Our in vitro studies illustrate that methotrexate can suppress SARSCoV-
2 entry and replication by targeting furin and DHFR of the host,
respectively. Additionally, methotrexate inhibits all four SARS-CoV-2 variants
of concern. In a Syrian hamster model for COVID-19, methotrexate reduced
virus replication, inflammation in the infected lungs. By analysis of
transcriptomic analysis of collected samples from hamster lung, we
uncovered that neutrophil infiltration and the pathways of innate immune
response, adaptive immune response and thrombosis are modulated in the
treated animals.
Conclusions: We demonstrate that this systematic repurposing approach is
potentially useful to identify pharmaceutical targets, multi-target drugs and
regulated pathways for a complex disease. Our findings indicate that
methotrexate is established as a promising drug against SARS-CoV-2 variants
and can be used to treat lung damage and inflammation in COVID-19,
warranting future evaluation in clinical trials.