陳亭妏助理教授研究團隊發表研究成果於 Journal of Infection
連結網址:https://www.sciencedirect.com/science/article/pii/S0163445322005175?via%3Dihub
Summary
Objectives: RNA therapeutics is an emerging field that widens the range of treatable targets and would improve disease outcome through bypassing the antibiotic bactericidal targets to kill Mycobacterium tuberculosis (M.tb).
Methods: We screened for microRNA with immune-regulatory functions against M.tb by next generation sequencing of peripheral blood mononuclear cells, followed by validation in an independent cohort.
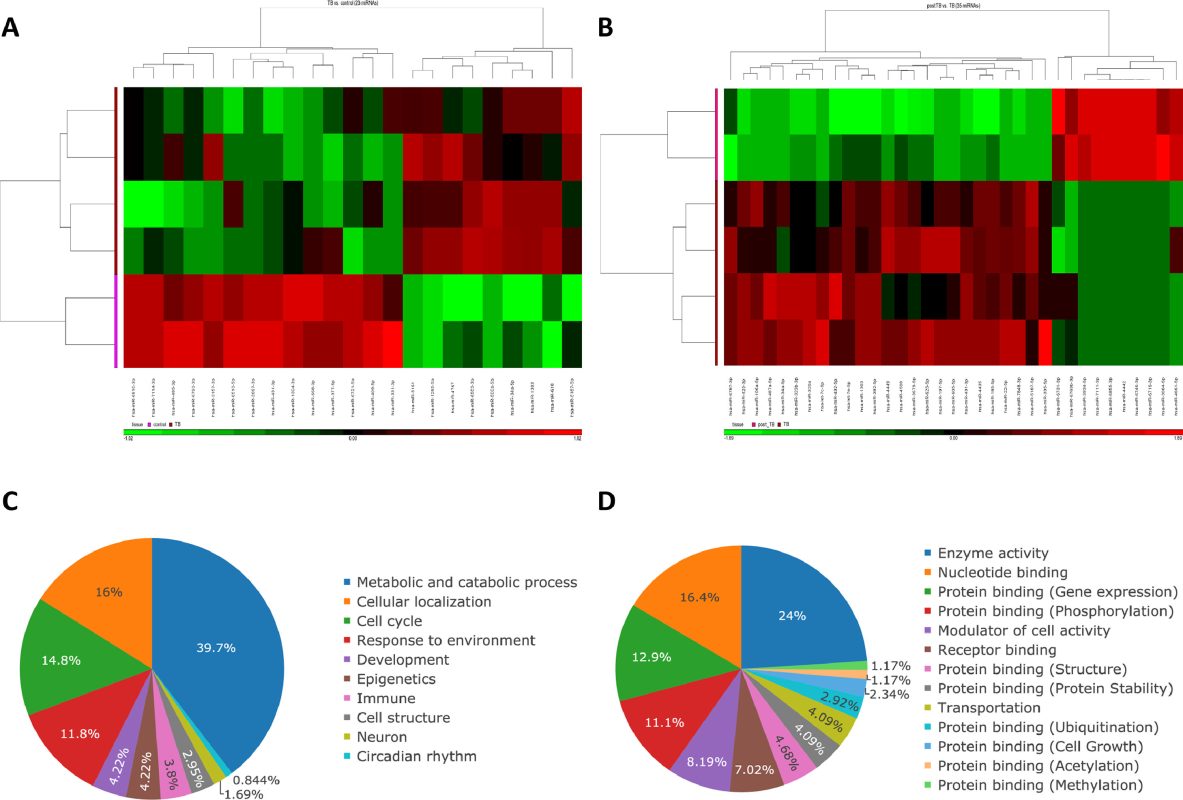
Results: Twenty three differentially expressed microRNAs were identified between 12 active pulmonary TB patients and 4 healthy subjects, and 35 microRNAs before and after 6-month anti-TB therapy. Enriched predicted target pathways included proteoglycan, HIF-1 signaling, longevity-regulating, central carbon metabolism, and autophagy. We validated miR-431–3p down-regulation and miR-1303 up-regulation
accompanied with corresponding changes in their predicted target genes in an independent validation cohort of 46 active TB patients, 30 latent TB infection subjects, and 24 non-infected healthy subjects. In vitro experiments of transfections with miR-431–3p mimic/miR-1303 short interfering RNA in THP-1 cells under ESAT-6 stimuli showed that miR-431–3p and miR-1303 were capable to augment and suppress autophagy/apoptosis/phagocytosis of macrophage via targeting MDR1/MMP16/RIPOR2 and ATG5, respectively.
Conclusions: This study provides a proof of concept for microRNA-based host-directed immunotherapy for active TB disease. The combined miR-431–3p over-expression and miR-1303 knock-down revealed new vulnerabilities of treatment-refractory TB disease.